Description
| Stock Solution | ||
| Carmine | 2.5 | g |
| Potassium carbonate | 1.2 | g |
| Ammonia | 25 | mL |
| Distilled water | 75 | mL |
| Combine the water, potassium carbonate and carmine. Boil for 15 minutes. Cool and filter. Add the ammonia. |
| Working solution | ||
| Stock solution | 25 | mL |
| Methanol, absolute | 35 | mL |
| Ammonia | 35 | mL |
| Differentiator | ||
| Ethanol, 95% | 40 | mL |
| Methanol, absolute | 20 | mL |
| Distilled water | 50 | mL |
Tissue sample
5µ paraffin sections of neutral buffered formalin fixed tissue are suitable. It is often recommended that an alcoholic fixative be used to preserve glycogen, but the majority may be demonstrated following formalin fixation. Alcohol fixation is not usually required except for critical applications.
Method
- Bring sections to water via xylene and ethanol.
- Stain nuclei with hemalum. It is not necessary to blue.
- Rinse with distilled water.
- Place in the working solution overnight.
- Rinse well with water.
- Place in the differentiator until glycogen granules are prominent.
- Dehydrate with ethanol.
- Clear with xylene and mount with a resinous medium.
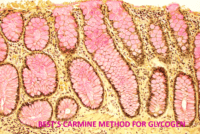
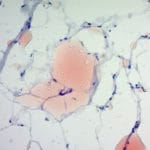
Expected results
- Glycogen – red
- Nuclei – blue
Notes
- Because of the high alcohol content the working solution should be filtered and used in a covered Coplin jar.
- The solutions are strongly alkaline and may remove sections from slides. Ensure sections are well baked on.
- The stock solution slowly deteriorates. If it fails to stain within the usual time, a fresh solution should be made.
- The demonstration of glycogen with this method is probably due to hydrogen bonding of the carmine to the glycogen. The potassium chloride and ammonia suppressing background staining by the carmine.
- The method is not as popular as in the past since PAS diastase clearly identifies glycogen, albeit negatively.
Reference
Gray, Peter. (1954)
The Microtomist’s Formulary and Guide.
Originally published by:– The Blakiston Co.
Republished by:– Robert E. Krieger Publishing Co.
Citing:–
Best, (1906)
Archiv für mikroskopische Anatomie, v. 23, p. 520
Bonn, Germany
Bancroft, J. D. and Stevens, A., (1977)
Theory and practice of histological techniques
Churchill Livingstone, Edinburgh, UK
more informations see data sheet below




Reviews
There are no reviews yet.